Press Releases of the Week
|
|
|
|
|
 Elizabeth Nanouris Named VP of Business Operations at InFocus Clinical Research Elizabeth Nanouris Named VP of Business Operations at InFocus Clinical Research
InFocus Clinical Research, the only global, full-service retina specialty contract research organization, announced that Elizabeth Nanouris has joined InFocus as vice president of business operations…
|
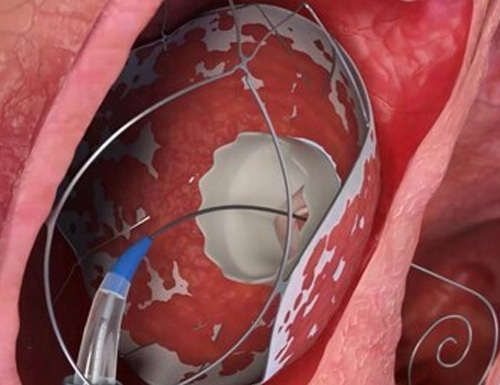 TruLeaf Medical Completes Second-Stage Valve Implantation in Two Patients TruLeaf Medical Completes Second-Stage Valve Implantation in Two Patients
TruLeaf Medical, Ltd, a fully-owned subsidiary of Allmed Solutions (TASE: ALMD), a clinical-stage medical device company specializing in transcatheter structural heart innovations, is pleased to…
|
 Novo Nordisk’s Ozempic pill—the only FDA-approved oral peptide GLP-1 for adults with Type 2 Diabetes—will soon be available in the U.S Novo Nordisk’s Ozempic pill—the only FDA-approved oral peptide GLP-1 for adults with Type 2 Diabetes—will soon be available in the U.S
Novo Nordisk announced that starting Monday, May 4, Ozempic® (semaglutide) tablets 1.5 mg, 4 mg, and 9 mg will be available for adults with type 2 diabetes in the US. Ozempic® is the only FDA…
|
 Incyte announces FDA approval of Jakafi XR for treating myelofibrosis, polycythemia vera, and graft-versus-host disease Incyte announces FDA approval of Jakafi XR for treating myelofibrosis, polycythemia vera, and graft-versus-host disease
Incyte announced that the U.S. Food and Drug Administration (FDA) has approved Jakafi XR™ (ruxolitinib) extended-release tablets for the treatment of adults with intermediate- or high-risk…
|
 UCB to acquire Candid Therapeutics, strengthening its immunology pipeline with novel T-cell engagers UCB to acquire Candid Therapeutics, strengthening its immunology pipeline with novel T-cell engagers
UCB, a global biopharmaceutical company, announced signing of a definitive agreement under which it would acquire Candid Therapeutics (Candid), a privately held clinical-stage biotechnology company…
|
|
|
 Pharmacists gain a flexible, space-saving option as YARAL Pharma launches its 15-count Diclofenac Epolamine Topical System 1.3% Pharmacists gain a flexible, space-saving option as YARAL Pharma launches its 15-count Diclofenac Epolamine Topical System 1.3%
YARAL Pharma Inc. the rapidly growing U.S. generics subsidiary of IBSA (Institut Biochimique SA), announced that its Diclofenac Epolamine Topical System 1.3%, the authorized generic of Flector®…
|
 Lasix® ONYU® Receives CE Marking, Advancing Toward International Authorization; EMA Approves for Centralized Review Based on Innovation Lasix® ONYU® Receives CE Marking, Advancing Toward International Authorization; EMA Approves for Centralized Review Based on Innovation
SQ Innovation, developer of Lasix® ONYU, announced two important regulatory milestones in its international expansion strategy: the receipt of a CE marking certificate for the Lasix ONYU device, and…
|
 Rapid Nexus Secures VA and DoD Contracts, Launches Commercial Deployment of FDA-Cleared Wound Technology Rapid Nexus Secures VA and DoD Contracts, Launches Commercial Deployment of FDA-Cleared Wound Technology
Rapid Nexus Nanotech Wound Solutions, Inc., a California-based medical technology company, announced it has secured supply contracts with the U.S. Department of Veterans Affairs (VA) and Department…
|
 Apertura Gene Therapy and Broad Institute scientists share data on TfR1 CapX™, a novel CNS-targeting capsid Apertura Gene Therapy and Broad Institute scientists share data on TfR1 CapX™, a novel CNS-targeting capsid
Apertura Gene Therapy, a biotechnology company developing next-generation AAV capsids for delivering genetic medicines, along with scientists from the Broad Institute of MIT and Harvard, will present…
|
|
|
|